What is an example of a metal activity series practice problem?
1 Answer
Jun 24, 2018
Here is a common question with single replacement reactions:
Explanation:
Given:
Well, let's see:
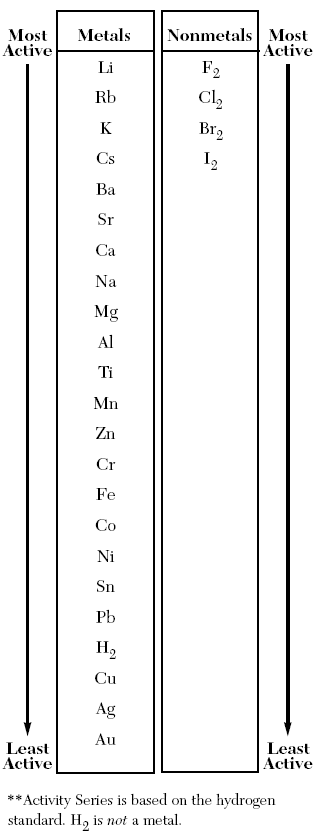
In this equation, we need to see if
If an element is
So, is Zn higher than H?
Therefore, this reaction can happen.
Another question may ask something like:
Which element can NOT replace
The answer has to be something lower on the table (less active) than Hydrogen. The only viable answers are
Hope this helps!

