What is an example of a tetrahedral bent molecule other than water?
1 Answer
Apr 27, 2016
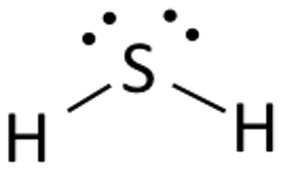
Since sulfur atom is larger than oxygen atom (it has a new quantum level), the
#vecr_("H"-"S") = "133.6 pm"#
#vecr_("H"-"O") = "95.8 pm"#
Thus, the
Therefore, we can expect

