What is most of the matter around you is in the form of (elements, compounds)?
1 Answer
Oct 4, 2016
Most of the matter around you and inside you is made up of compounds.
Explanation:
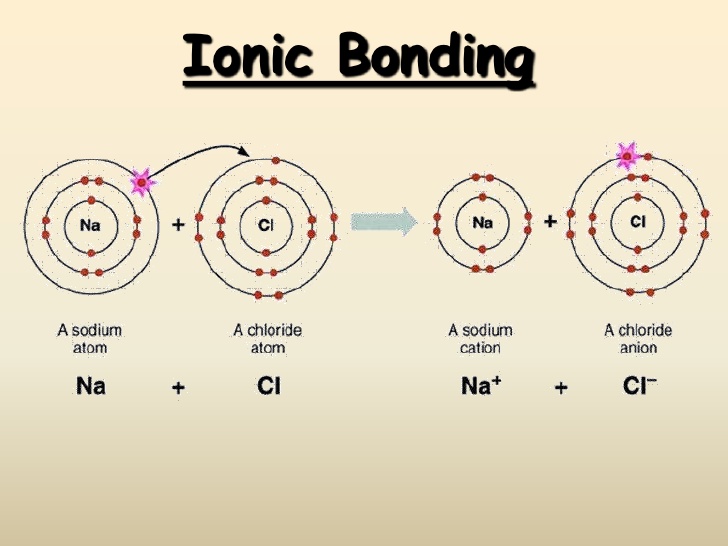
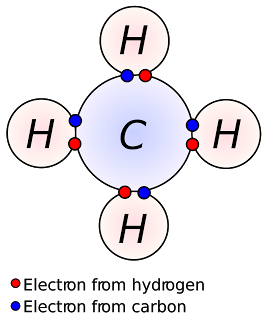
Most elements are not stable in their elemental form. In order to become stable, they chemically combine with atoms of other elements through covalent and ionic bonding. Covalent bonding involves sharing electrons, usually between nonmetals, and ionic bonding involves the transfer of electrons from one atom to another, primarily between metals and nonmetals or metals and polyatomic ions.
Example of covalent bonding
Example of ionic bonding