What is quantum dot light?
1 Answer
Quantum dots (QDs) are semiconductor particles, typically on the order of a few
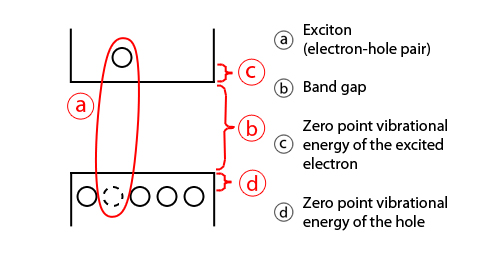
The following is a simplified energy diagram for a quantum dot, showing its band gap, or HOMO-LUMO gap (more on the HOMO-LUMO gap is described here):
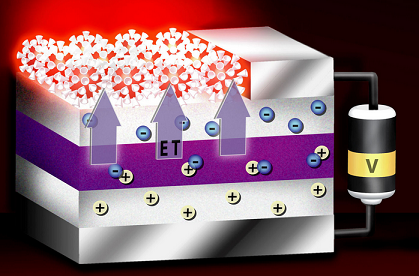
The idea is that applied electric fields will draw electrons (solid circles,
#""_(-1)^(0) e + ""_(+1)^(0) e -> 2 ""_(0)^(0) gamma#
Or if you prefer, a diagram:

[Read more about this here.]
Depending on the dot size, they can be made to emit different colors of light; the wavelength emitted is directly proportional to the QD size.

This relationship can be seen by modeling semiconductors using the one-dimensional Particle in a Box energy levels:
#E_n = (n^2h^2)/(8m_eL^2)# where:
#h = 6.626 xx 10^(-34) "J"cdot"s"#
#m_e = 9.109 xx 10^(-31) "kg"#
#L# is the length of the "box".
#n = 1, 2, 3, . . . # is the principal quantum number.
So, since
Common applications for them are in transistors, solar cells, and quantum computing. You can find out how they are made by looking here.

