What is reductive elimination in elimination reactions?
1 Answer
DISCLAIMER: You will NOT see this in first-semester inorganic chemistry.
This can be seen is in the elimination of two ligands from an octahedral metal-ligand complex, reducing the metal to a lower oxidation state.
In general, the reduction of the transition metal in an octahedral complex reduces the number of electrons around it by 2, from 18 to 16.
This is the opposite of oxidative addition.
EXAMPLE 1
One example is the concerted reduction of
I've highlighted the ligands that leave the octahedral complex to form the new square planar complex. Afterwards, the chlorine, which was previously axial, has shifted to take the place of the now-missing equatorial methyl and becomes equatorial.
Furthermore, this complex reduces from 18 to 16 electrons around the central metal.
Since the
Then, after the two hydrides leave, rhodium reduces to a
EXAMPLE 2
Another example is the concerted reduction of
Notice how both of these concerted reductive elimination reactions required a cis relationship to the to-be-eliminated ligands. The acyl didn't leave with the chloride in the first reaction, nor did the hydride leave with the carbonyl in the second reaction.
As before, this complex also reduces from 18 to 16 electrons around the central metal.
Since the two
Then, after the two hydrides leave, we gain a
The mechanism for this seems to be the following:
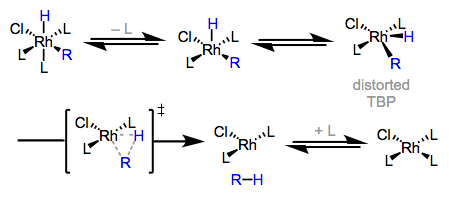
- The removal of the axial ligand to form a square pyramidal intermediate.
- The realignment of the remaining ligands into a distorted trigonal bipyramidal complex.
- The interaction of the to-be-eliminated ligands with the transition metal in a transition state (marked double-dagger)
- The elimination of the aforementioned ligands.
- The re-addition of the ligand removed in step 1.
I would not expect that the curved-arrow mechanism is known with good certainty, so don't feel like you have to figure out how to draw the mechanistic arrows.

