What is the difference between enantiomers and chiral molecules?
2 Answers
Mar 7, 2018
No difference
Explanation:
Chiral molecules are molecules that, having 1 or more asimmetric tetraedric centers, exist as 2 or more enantiomers
Mar 7, 2018
Enantiomers are a subset of chiral molecules.
Explanation:
Enantiomers
Enantiomers are chiral molecules that are nonsuperimposable mirror images of each other.
For example, D-glucose and L-glucose are enantiomers.

Chiral molecules
A chiral molecule is a molecule that is not superimposable on its mirror image.
For example, here are two chiral molecules.
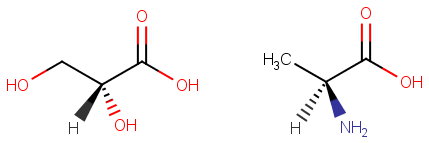
They are chiral, but they are not enantiomers.
There are thousands of chiral compounds, but only those that are nonsuperimposable mirror images of each other are enantiomers.


