What is the Lewis structure of Cl_3?
1 Answer
May 13, 2018
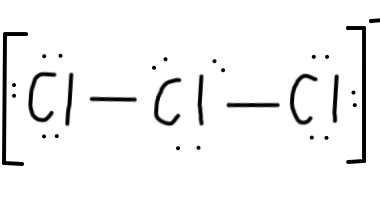
Explanation:
(Assuming that
- The total valence electrons for
Cl_3^- is7xx3+1=22 , sinceCl has7 valence electrons and there's a negative charge (which adds1 electron). - Let's draw a single bond between the
3 Cl atoms to begin:
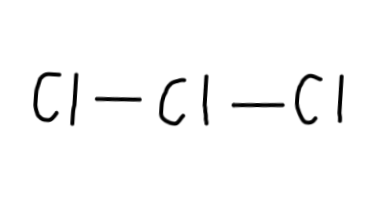
Now, we have22-4=18 electrons left to put on the diagram, since each single bond counts as2 electrons. - Then, we should complete the octets for all of the
Cl atoms:
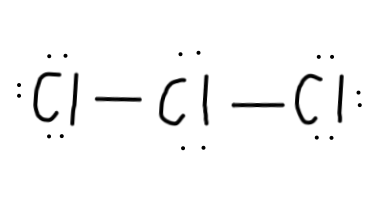
- We still have
2 electrons left after placing all those valence electrons.Cl can have an expanded octet, though, so that's not too much of a problem. - To determine where to put this extra electron pair, let's calculate the formal charges of all the situations:
- If the extra electron pair is placed on the left
Cl atom, then the formal charge on thatCl atom would be7-9=-2 . - If it were placed on the right
Cl atom, then thatCl atom would have a formal charge of7-9=-2 . - If it were placed on the central
Cl atom, then thatCl atom would have a formal charge of7-8=-1 . - So, the best placement for that extra electron pair would be the central
Cl , since that arrangement minimises formal charge. - Let's draw that:
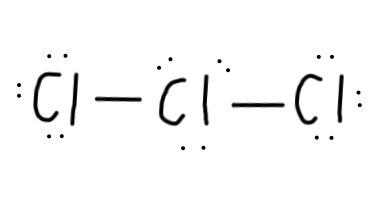
- And add a bracket around everything, with a negative sign, to indicate that this is a negatively charged ion:
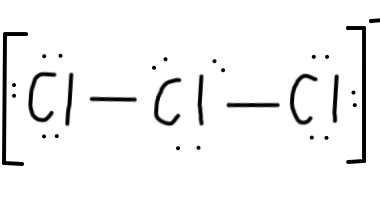
- Finally, let's do a last double-check for the number of electrons. We should end up with
22 electrons. :)

