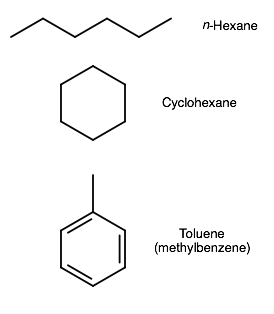
What is the molecular formula for hexane, cyclohexane, and toluene?
1 Answer
Nov 20, 2015
You can easily look these up, but let's just figure it out without drawing it first...
n-Hexane has six carbons and is a saturated hydrocarbon. It is the most simple compound of the three. As a straight-chained alkane, it has the general formula
Cyclohexane is simply the same compound as a ring, giving it a general formula of
Toluene is the "common" name for methylbenzene. Benzene is