Drawing Cyclohexanes in Boat and Chair Conformations
Key Questions
-
Answer:
The chair conformation is special because it is the most stable arrangement of atoms in the cyclohexane ring.
Explanation:
We tend to think of cyclohexane as a planar ring, as in the diagram below.
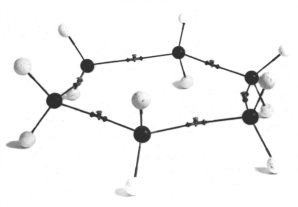
(from www.hyle.org)But this would be a high-energy structure. The bond angles would be 120° instead of 109.5°, and all the H atoms would be eclipsed.
Some of the angle strain can be relieved if one of the C atoms is lifted out of the ring to form a half-chair.
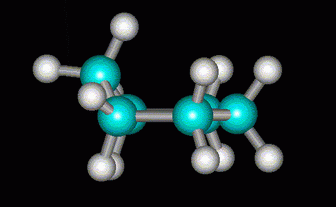
(from course1.winona.edu)More strain would be relieved if the C atom at the other end were also lifted to form a boat.
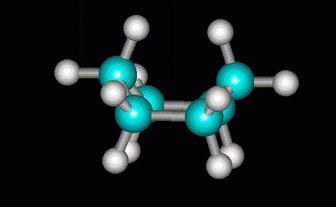
(from course1.winona.edu)But this introduces severe steric hindrance between two of the H atoms, and the H atoms on the sides are all eclipsed.
If the C atom is bent the other way, we get the chair form of cyclohexane.
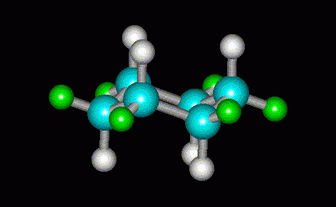
(from course1.winona.edu)There is no angle strain, and the H atoms are all staggered.
The chair is the most stable form of cyclohexane.
There are actually two forms of the cyclohexane chair.

(from classes.yale.edu)At room temperature, they interconvert about 100 000 times per second.
-
Answer:
Here's the method I use to draw cyclohexane boats.
Explanation:
A cyclohexane boat is like boat — it has a bow, a stern and two sides.
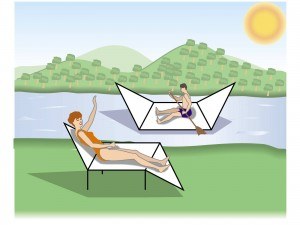
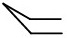
This is how you draw a boat.
Step 1. Draw two horizontal parallel lines.

These will form the sides of the boat.
Step 2. Add two lines pointing up to make the bow of the boat.
Step 3. Add two lines pointing up at the other end to make the stern.
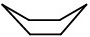
And your boat is complete!
-
Answer:
Here's the method I use to draw cyclohexane chairs.
Explanation:
A cyclohexane chair is like an outdoor lounge chair.
It has a seat, a backrest, and a leg rest.
I'll show you how to draw both chairs at the same time.
Step 1. Draw two horizontal parallel lines, one slightly offset from the other.

These will form the seat of the chair.
Step 2. Add two lines pointing up to make the backrest of the chair.

Step 3. Add two lines pointing down to make the leg rests.

And your chair is complete!