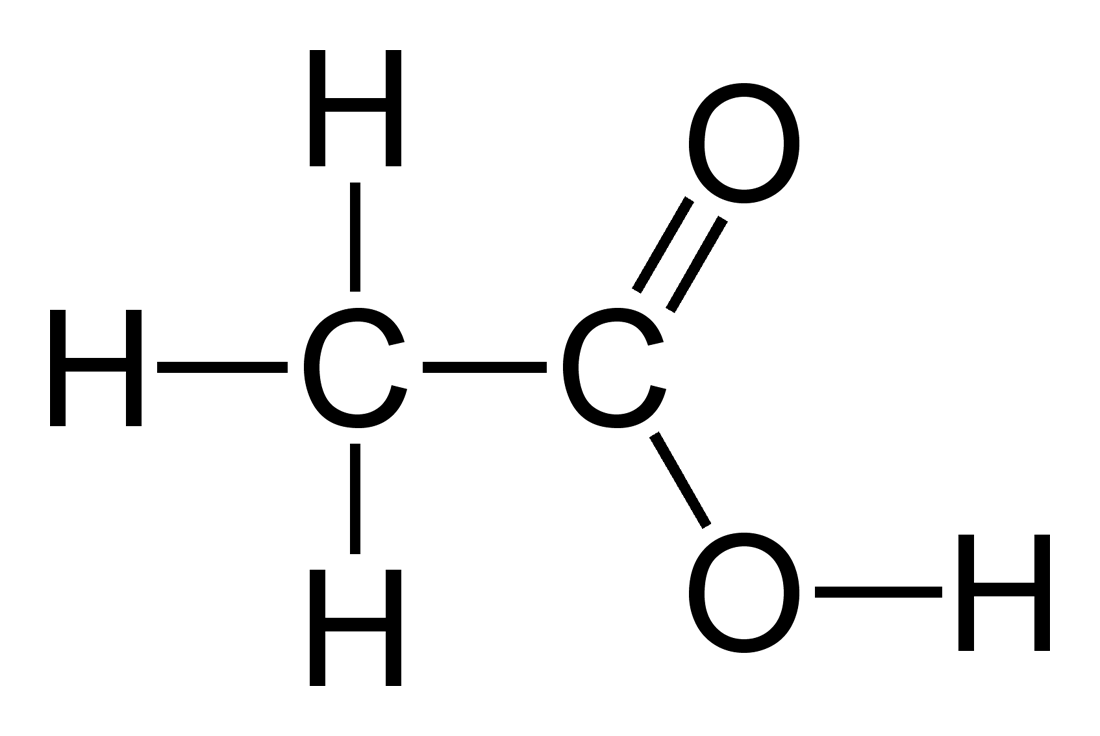
What is the old and new chemical name for vinegar and name the family of salts obtained from this acid?
1 Answer
Common name: Acetic acid
Systematic IUPAC name: Ethanoic acid
Vinegar: a diluted mixture of water and acetic acid.
Acetate salts are called acetates or ethanoates
Explanation:
The word "vinegar" comes from the french word " vin aigre" meaning "sour wine. It was used to describe wines that had gone bad and started to taste gross. Vinegar used today is a diluted mixture of acetic acid
The International Union of Pure and Applied Chemistry (IUPAC) sets nomenclature rules for all chemical substances to maintain consistency. Ethanoic acid is more commonly known by its common name of acetic acid . While both are equivalent, you may be asked for the systematic IUPAC name on an exam, in which case you would give ethanoic acid.
Students in general chemistry will often be asked to memorize a set of polyatomic ions, in which the acetate ion
When you get to organic chemistry, you will learn that acetic acid has the