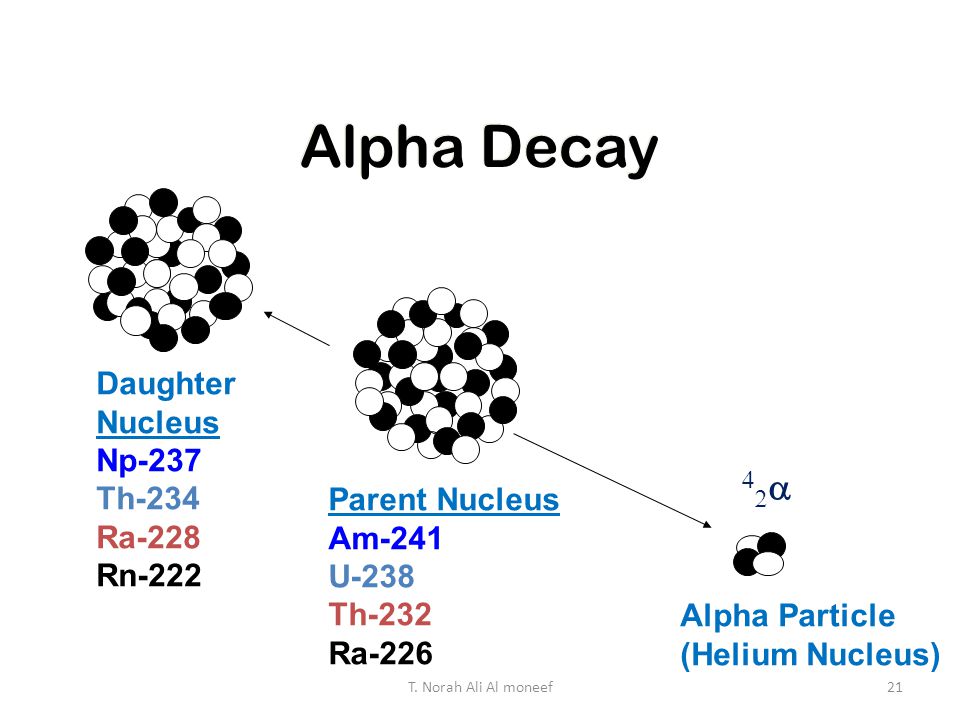
What is the product produced when #""_92^238"U"# undergoes alpha decay?
1 Answer
May 17, 2018
Thorium-
Explanation:
The equation for alpha decay is:
where:
So here, the new mass number of the atom will be
So, the equation for the alpha decay of uranium is: