What is the reducing agent in the reaction 2Na + 2H_2O -> 2NaOH + H_22Na+2H2O→2NaOH+H2?
1 Answer
Sodium metal.
Explanation:
You're dealing with a redox reaction in which sodium metal,
2stackrel(color(blue)(0))("Na")_ ((s)) + 2stackrel(color(blue)(+1))("H")_ 2 stackrel(color(blue)(-2))("O")_ ((l)) -> 2stackrel(color(blue)(+1)) ("Na") stackrel(color(blue)(-2))("O") stackrel(color(blue)(+1))("H")_ ((aq)) + stackrel(color(blue)(0))("H") _(2(g))20Na(s)+2+1H2−2O(l)→2+1Na−2O+1H(aq)+0H2(g)
As you can see, the oxidation state of sodium goes from
On the other hand, the oxidation state of hydrogen goes from
Now, a reducing agent is responsible with reducing a chemical species that takes part in a redox reaction. Similarly, an oxidizing agent is responsible with oxidizing a chemical species that takes part in a redox reaction.
You can thus say that the chemical species that is being oxidized acts as a reducing agent for the chemical species that is being reduced.
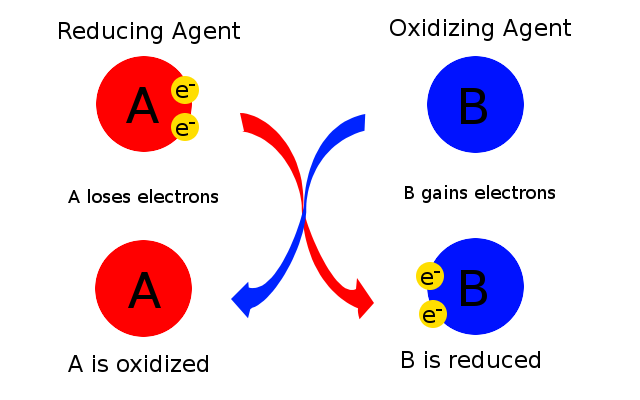
In this case, sodium metal is being oxidized, which implies that it is acting as the reducing agent.

