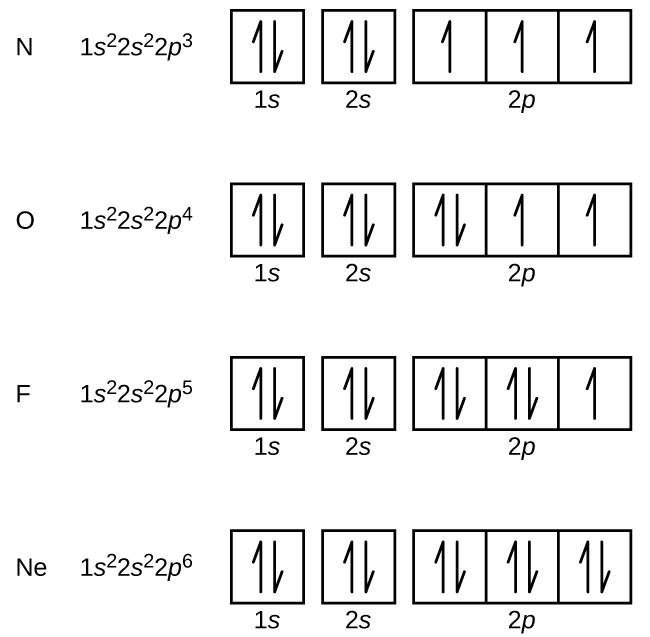
When electrons ocoupy different orbitals of the same sub-level, do they all have the same spin?
1 Answer
Sep 9, 2017
Only as single electrons in the orbitals of a partially filled sublevel.
Explanation:
An orbital can contain a maximum of two electrons with opposite spins. Each orbital in the same sublevel must have one electron with the same spin before a second electron with an opposite spin enters the orbital. When there are multiple orbitals in the same sublevel, such as the three