Where do gases dissolve most easily?
2 Answers
In a solvent with similar molecular properties.
Explanation:
A popular aphorism used for predicting solubility is "like dissolves like." Which means that polar solvents are good at dissolving polar solutes, and non-polar solvents are good at dissolving non-polar solutes.
Here are some examples:
- Ammonia (a highly polar molecule) dissolves well in water (which is a common polar solvent.)
Ammonia Lewis dot structure, source: Michael Blaber
- Most Halogens (excluding fluorine,
#F_2# ,) dissolve well in carbon tetrachloride#C Cl_4# (a non-polar solvent) while hardly dissolve in water
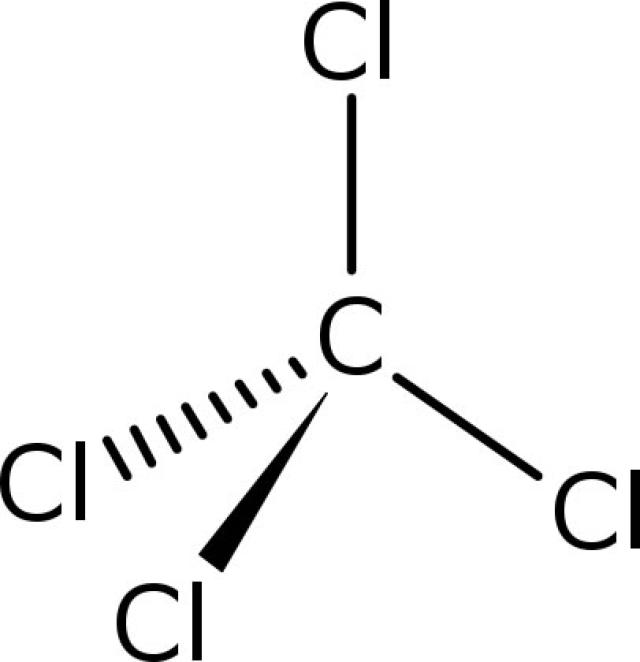 Molecular structure of carbon tetrachloride, its tetrahedral shape cancels out the polarity in the #C-Cl# bonds.
Molecular structure of carbon tetrachloride, its tetrahedral shape cancels out the polarity in the #C-Cl# bonds.
Gases dissolve must easily in solutions that are under pressure and at low temperatures.
Explanation:
Take Soda water for an example. A common experience is drinking an ice cold coke. The solution of carbon dioxide with the liquid (mainly water) is most concentrated when the liquid is cold and under pressure. Relive the pressure by opening the bottle and bubbles of Carbon Dioxide come rapidly to the surface. This shows that gases dissolves most easily when under pressure.
Another common experience is going back to drink a can of coke that has been opened for a while and left in the sun. The soda is flat there is little or no carbon dioxide still left in solution. This shows that gases dissolve best in a cold solution not in a warm solution.
Another example is Oxygen in rivers and lakes. During the summer warm water has less Oxygen gas dissolved in the water.
So gases dissolve must easily in solutions that are under pressure and at a cold temperature.

