Which low-energy state of condensed matter is characterized by structural rigidity and resistance to changes of shape or volume?
1 Answer
It's a solid.
Explanation:
The particles that make up a solid have the lowest energy, and vibrate in place. This results in the characteristics of a solid, such as a definite shape and volume. A solid is actually the frozen state of matter for a substance. A substance does not have to freeze at a cold temperature. For example, the faucet you have at your lab station in chemistry, and the faucets in your home are frozen stainless steel; and so is your desk, pencils, pens, cell phones, and computers are all frozen. Anything solid is frozen.
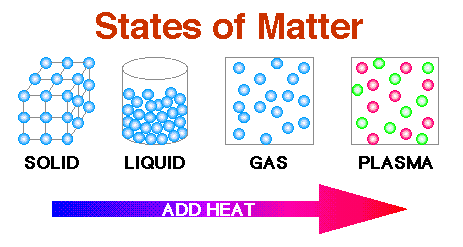
The diagram below shows that by adding heat, which increases the energy and speed of the particles, the solid melts into the liquid state, then as more energy is applied, the liquid becomes a gas, and if enough energy is added, it will become plasma.
https://en.m.wikipedia.org/wiki/State_of_matter
 http://www.youcanlearn.com/seven-incredible-concepts-in-physical-science/
http://www.youcanlearn.com/seven-incredible-concepts-in-physical-science/

