Which of the following molecules may have hydrogen bonds between other molecules of the same kind? CH3F, CH3NH2, CH3OH, CH3Br
1 Answer
Aug 6, 2014
Only CH₃NH₂ and CH₃OH
Explanation:
Only CH₃NH₂ and CH₃OH can have hydrogen bonds between other molecules of the same kind.
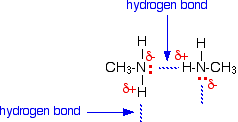
To have hydrogen bonding, you need an N, O, or F atom in one molecule and an H attached to an N, O, or F atom in another molecule.
CH₃F has an F atom, but no H-F bonds It cannot form hydrogen bonds with other CH₃F molecules.
CH₃NH₂ has an N atom and N-H bonds It can form hydrogen bonds with other CH₃NH₂ molecules.
CH₃OH has an O atom and an O-H bond. It can form hydrogen bonds with other CH₃OH molecules.
CH₃Br has no N, O, or F atoms, and it has no H atoms attached to N, O, or F. It cannot form hydrogen bonds with other CH₃Br molecules.