Why do molecules form bonds?
1 Answer
Molecules usually do not form bonds
Explanation:
Molecules are composed of atoms that have bonded and released the extra energy with them as bond formation energy.This bond can either be ionic or covalent.
But, sometimes molecules form coordinate bonds.This results in a complex, a compound formed due to coordinate bonding between two molecules.For ex, ammonia-boron triflouride complex
[
It is formed when nitrogen of
Sometimes two covalent molecules form a special type of bond called bridge bond and exist as diamers for example
In bridge bonding of
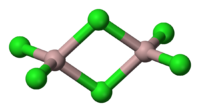
There are several other cases in which molecules form bonds and all of which I cannot discuss here.
In conclusion, molecules form bonds to achieve stability by filling up empty orbitals or by neutralizing charge as in hydrogen bonds.

