Polar Protic, Polar Aprotic and Non-Polar Solvents
Key Questions
-
Answer:
Non-polar solvents are non-polar molecules that can be used as solvent.
Explanation:
Non-polar solvents are any non-polar molecules that can be used as a solvent.
Example:
Hexane, pentane, heptane, etc.
Carbon tetrachloride"C"Cl_4CCl4 . -
Here's an example.
Let's hypothetically react
Li^((+)) [(CH_2)_3CH_3]^((-))Li(+)[(CH2)3CH3](−) (commonlyBuLiBuLi ) with acetone. Normally,BuLiBuLi is a fantastic nucleophile due to lithium's lewis acid characteristics.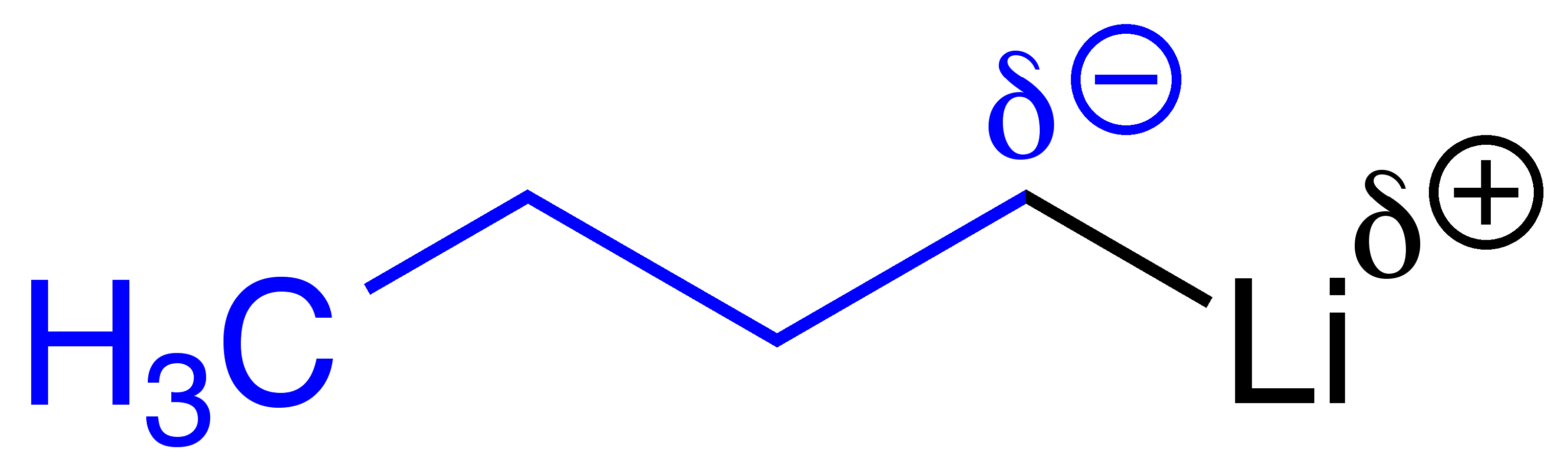
If you solvate
BuLiBuLi in the optimal amount of ethanol (commonlyEtOHEtOH ), you have now in solution, before anything happens,BuLiBuLi ,EtOHEtOH , and acetone.Acetone:
What would most likely happen is that since
BuLiBuLi has such a high nucleophilicity, instead of reacting with acetone all the time, there is a good chance it would also steal a proton fromEtOHEtOH .At that point,
BuLiBuLi would become butane, which is clearly nonreactive as a poor nucleophile. Then,EtO^(-)EtO− forms and it becomes a potential nucleophile to attack acetone (but less often, as it's a worse nucleophile).At this point, you may realize that you now have a situation where:
BuLiBuLi grabs a proton and loses its reactivity, allowingEtO^(-)EtO− to be an additional nucleophile (there's still someBuLiBuLi leftover)BuLiBuLi attacks acetone and the reaction proceeds toEtOHEtOH protonating the tetrahedral intermediate to form a tertiary alcohol.
The result then is a mixture of the butane,
EtOHEtOH , acetone, the tertiary alcohol, and the product of the mechanism whereEtO^(-)EtO− attacks acetone. Ideally you don't want a mixture that you'd have to separate and purify later. If you got a pure product, that's what you should want.So naturally, it's a good idea, for example, to not use a protic solvent when using an anionic nucleophile, because it may actually deactivate the nucleophile.