Choosing between E1 and E2
Key Questions
-
E1 reactions are unimolecular whereas E2 reactions are bimolecular. By extension,
#R_("E"1) = k["LG"^-]# , and#R_("E"2) = k["LG"^-]["NUC"]# where the notations stand for leaving group and nucleophile.
As a consequence of the preceding, E2 reactions usually proceed with a strong nucleophile (e.g. base), whereas E1 reactions are fine with a weaker nucleophile (e.g. base).
Mechanistically, E2 reactions are concerted (and occur faster), whereas E1 reactions are stepwise (and occur slower and at a higher energy cost, generally).
Due to E1's mechanistic behavior, carbocation rearrangements can occur in the intermediate, such that the positive charge is relocated on the most stable carbon. Factors influencing this include hyperconjugation and resonance. As a result of this, E1 usually won't occur on primary substrates.
Moreover, when competing with SN1, E1's rate is more sensitive and will dominate when supplied with heat.
If you'd like to see mechanisms, ask!
-
Answer:
#"E1"# eliminations follow Zaitsev's rule. With two exceptions,#"E2"# reactions also follow Zaitsev's Rule.Explanation:
(1) Certain cyclohexane derivatives.
Some cyclohexane derivatives, such as menthyl chloride, form the Hofmann product instead:
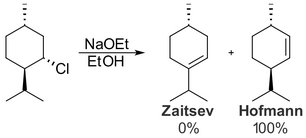
(from en.wikipedia.org)Formation of the Zaitsev product requires antiperiplanar elimination at the 2-position, but the isopropyl group – not the proton – is in the antiperiplanar location.
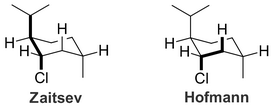
(from en.wikipedia.org)But there is an antiperiplanar hydrogen at the 6-position, so elimination occurs there and produces only the Hofmann product.
(2) Bulky bases
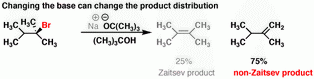
(from www.masterorganicchemistry.com)A bulky base attacks the least sterically hindered proton to form the Hofmann product.
-
E1 and E2 are both elimination reactions.
Both consist of a step where the leaving group leaves, and one where a proton is abstracted to form a
#pi# bond.Both can generally occur in polar protic solvent (there are extreme exceptions), and both are technically always in competition with nucleophilic substitution.
If you want mechanisms, just ask!