Question #d31d8
1 Answer
Sucrose is a dissacharide formed by the linkage of two monossacharides, glucose and fructose, and has the molecular formula

A Haworth projection is used to represent the cyclic structure of monossacharides in a simple 3D perspective. SInce sucrose is formed from one glucose and one fructose molecule, its Haworth projection will show an
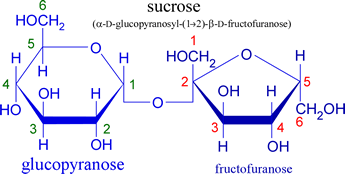
Now, a reducing sugar is a sugar that either has an aldehyde functional group attached, or is capable of forming one by way of isomerism. What basically takes place is a redox reaction in which the aldehyde is oxidized and another compound is reduced.
Another criterion that needs to be met in order to have a reducing sugar is that the anomeric carbon must be free to open up the ring structure and allow for the redox reaction to take place. A sugar must exist as the linear form in solution in order to be a reducing sugar.
In sucrose's case, both anomeric carbons, i.e. the one belonging to glucose (marked 1 in green in the above picture) and the one belonging to fructose (marked 2 in red), cannot open their respective rings because they are tied up in forming the glycosidic bond that keeps the sucrose molecule together.
As a result, sucrose cannot react with Benedict's reagent (basically a

