Question #67208
1 Answer
Phosphorus is located in group 15 of the periodic table and has an atomic number equal to 15. This means that a neutral phosphorus atom has a total of 15 electrons.
The ground state electron configuration for phosphorus is
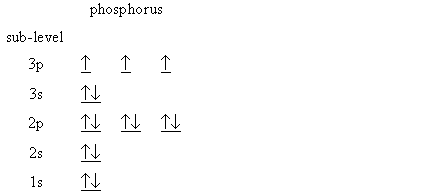
I'll use the noble gas shorthand notation from this point on.
Now, an excited state configuration has one electron promoted from a lower to a higher energy level. Looking at the ground state configuration, you could have one electron from the
Another possibility is to have one electron from the
Yet another possibility is to have one electron from the
Read more on how to determine excited state configurations here:
http://socratic.org/questions/how-do-you-determine-excited-state-electron-configuration

