Question #30d2b
1 Answer
Finding lone pairs using VSEPR Theory is a little atypical, since VSEPR Theory relies heavily on Lewis structures, which clearly show the lone pairs every atom that's part of a molecule has.
However, if you work backwards I guess you could determine how many (if any) lone pairs a central atom has. VSEPR Theory predicts two different geometries for a molecule: molecular geometry and electron geometry.
Let's take water, for example, and try to figure out how many lone pairs oxygen has. Here's water's molecular geometry
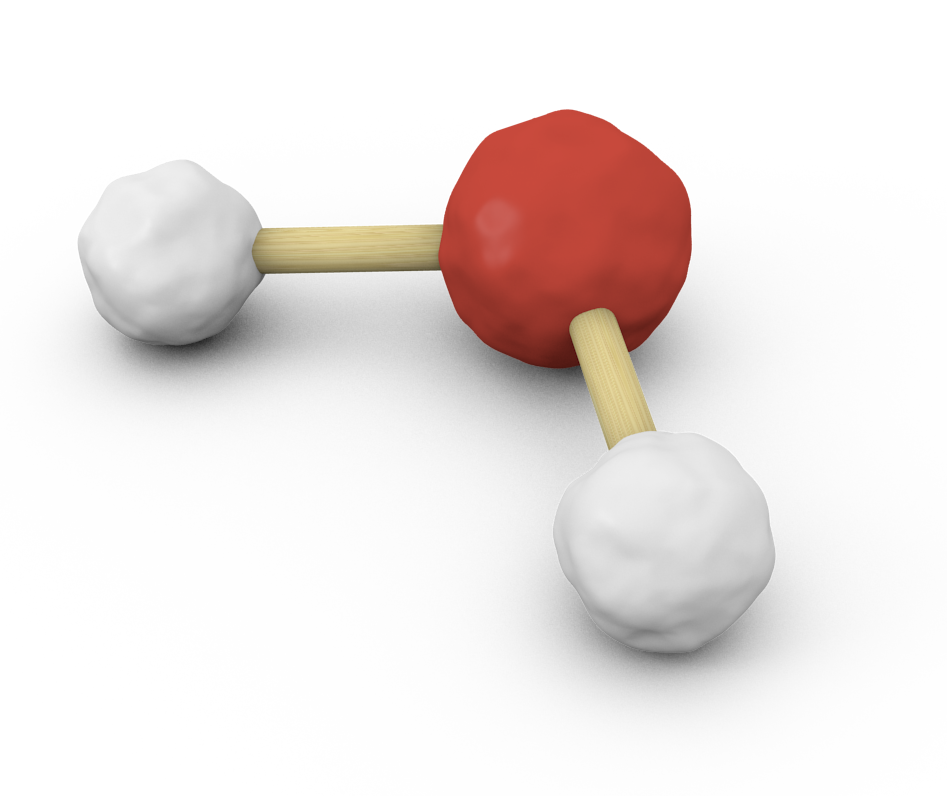
Notice the two hydrogen atoms connected to a central oxygen atom. Now, two important tools are used to determine a molecule's geometry: steric number (SN) and coordination number (CN).
The CN tells you how many atoms are bonded to, in this case, the central oxygen atom. In water's case, the CN will be equal to 2.
The SN tells you how many regions of electron density you have surrounding the oxygen atom. Regions of electron density can represent bonds with other atoms or lone pairs.
Now, if the SN is equal to the CN, the electron geometry of the molecule will be the same as its molecular geometry
Water's SN is 4, which implies that there are lone pairs around the oxygen atom. The two bonds oxygen has with the hydrogen atoms make up for 2 regions of electron density, which implies that you must have
Water's electron geometry is tetrahedral and looks like this:
As a conclusion, you could find out how many lone pairs you have on a central atom, but you must know the SN and the CN, or at least the molecular shape and the SN.


