Question #26b0e
1 Answer
The first thing you need to do when writing electron configurations is to determine exactly how many electrons your element or ion has.
Tin is located in period 5, group 14 of the periodic table, and has an atomic number equal to 50. That means that a neutral tin atom will have 50 electrons surrounding its nucleus.
Start with an electron configuration diagram like the one below (ignore the electrons already added)
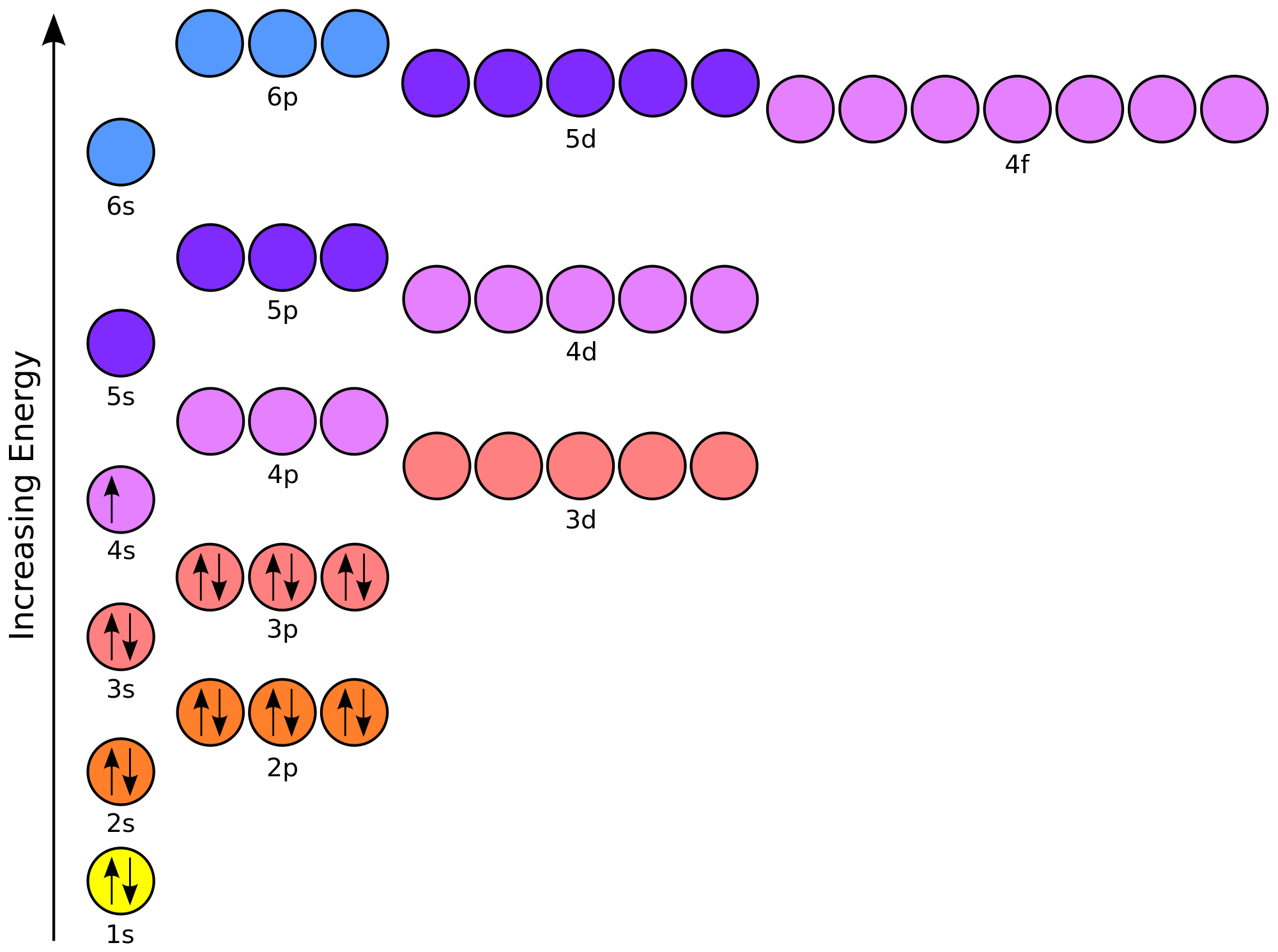
Add your electrons starting from the lowest energy orbital, the 1s-orbital, and move on up; remember to fill all orbitals located on the same energy level before moving up to a higher energy level.
You'd get
Moving on up
And there you have it - the electron configuration for tin is
As practice, you can try and name the element that has the electron configuration shown in the diagram I've used - notice that its configuration ends at

