Question #931d5
1 Answer
The phosphorus atoms in elemental phosphorus,
Explanation:
We know that elemental phosphorus has a tetrahedral structure.
Hence, the Lewis structure is

.
Each P atom has a lone pair and 3 bond pairs, for a steric number,
VSEPR theory predicts an
Compression from 109.5° to 60° would involve enormous angle strain.
If phosphorus used its unhybridized
More likely, each
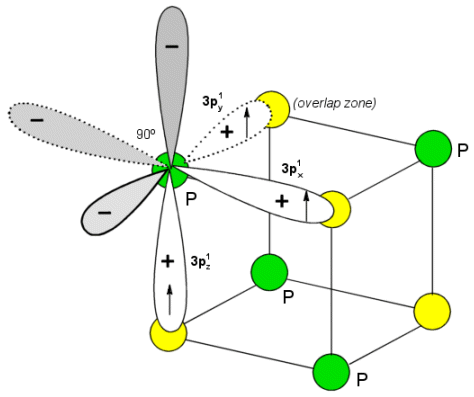
(Adapted from Chemexplore)
These bonds would not be as strong as ordinary σ bonds because the overlap angles are 90°.
Chemists usually call such bonds "bent bonds" or "banana bonds" because they are bent like a banana.
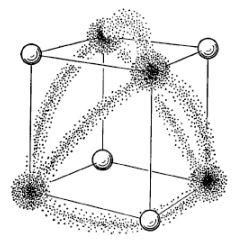
(Adapted from Chemexplore)

