Why is there greater repulsion between two lone pairs of electrons than between a lone pair and a bonding pair of electrons?
1 Answer
It's because the shape of the orbitals that hold lone pairs is different.
Explanation:
Indeed, the repulsion that exists between two lone pairs of electrons is greater than the repulsion that exists between a lone pair and a bonding pair of electrons.
Likewise, the repulsion that exists between a lone pair and a bonding pair of electrons is greater than the repulsion that exists between two bonding pairs of electrons*.
The reason behind why that is the case lies with the shapes of the orbitals that hold these long pairs.
Bonding electrons are located between two nuclei, which means that they have far less room to "move around" than lone pairs do.
A bond is essentially a region of very high electron density located between the nuclei of two atoms. This implies that the orbitals that hold these bonding electrons are more elongated than those that hold lone pairs.
By comparison, the orbitals that hold lone pairs are actually rounder and shorter than the orbitalas that hold the bonding electrons.
Here's a cool image showing how that would look like for a water molecule,

Lone pairs are located in orbitals that are more spherical than the orbitals in which bonding electrons reside.
Here's another example of how the lone pair of electrons on the ammonia molecule,
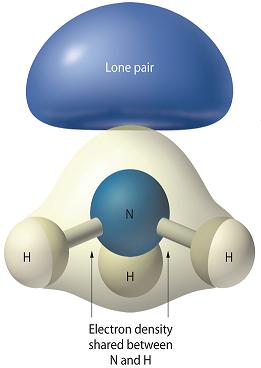
So, lone pairs of electrons take up more space than bonding electrons because they are only attracted by one nucleus, which implies that they more spread out at a shorter distance from that nucleus.
Bonding electrons are further away from the nucleus, but they are more localized, so they are not as spread out.
This is why two lone pairs will exhibit more repulsion that one lone pair and one bond pair, which in turn will exhibit more repulsion than two bond pairs.

