Question #68bfd
1 Answer
The electron doesn't move from one lobe to the other.
Explanation:
The electron is always in both lobes at once.
Half of the electron density is in one lobe, and half is in the other lobe.
The electron isn't a little white particle whizzing around the nucleus.
Instead, it behaves as a wave.
The node between the two lobes is just a node in its three-dimensional standing wave.
We can compare it with the second harmonic of a vibrating string.
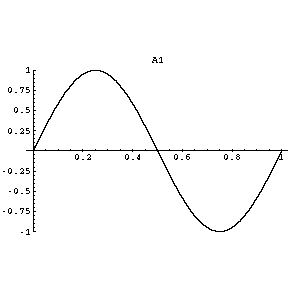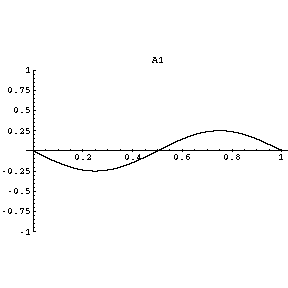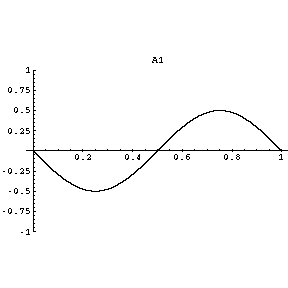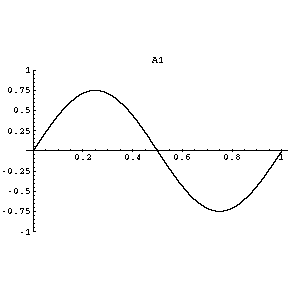
The two halves represent the two lobes of a
When the amplitude is positive in one node, it is negative in the other.
We could pinch the wave at the node, and the two halves would keep vibrating.
If you rotate the graph about the horizontal axis, you get a three-dimensional approximation to a
The standing wave in each lobe of a

