Question #93782
1 Answer
Here's what I get.
Here's the structure of cholesterol with the configuration at each chiral centre.
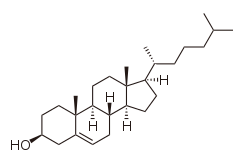
And here's another structure showing the numbering.

The eight chiral centres (with their attached groups in descending order of priority) are:
1.
∴ The configuration at
2.
The configuration at
3.
The configuration at
4.
The configuration at
5.
∴ The configuration at
6.
The configuration at
7.
The configuration at
8.
The configuration at
A complete stereochemical name would be
Now you see why we call it "cholesterol".

