Question #0491c
1 Answer
Sep 12, 2017
The sublevel that has the highest amount of energy of the given possibilities is
Explanation:
The
Refer to the image representing the Aufbau principle, which gives the order filling of sublevels according to the amount of energy. Each sublevel must be filled with electrons before filling the next higher energy level:
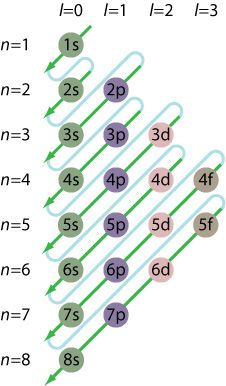
Note: The Aufbau principle is not dogma. About 20 transition elements do not follow the Aufbau exactly.

