Question #90e91
1 Answer
Nov 24, 2017
Explanation:
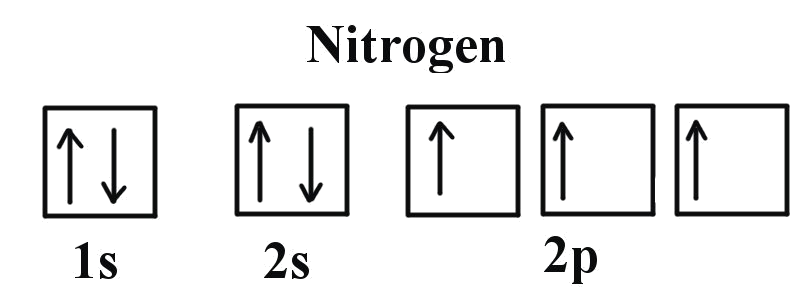
Valence shell electronic configuration of nitrogen is
Octet configuration is most stable electronic configuration.
So, to achieve stability it needs 3 electrons in
How do you write the noble-gas electron configuration for nitrogen?

