Question #0a7ec
1 Answer
Mar 15, 2017
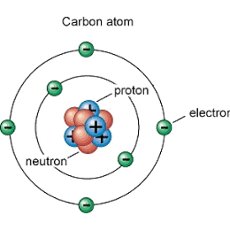
The number of valence electrons in a neutral carbon atom has four valence electrons, not eight. It is in group 14/IVA, not group 8. Only noble gases exist in nature with 8 valence electrons.
Explanation:
The electron configuration for a neutral carbon atom is