Question #883ac
1 Answer
(not balanced)
Explanation:
The diagram may be best:
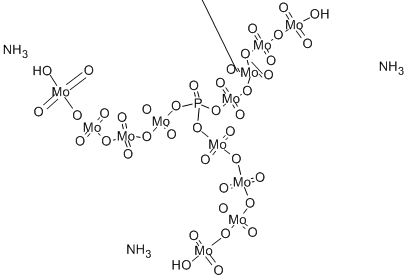
Graphic from http://www.chemicalbook.com/ChemicalProductProperty_EN_CB6162083.htm
Phosphate can form complexes with many di and tri-valent metals. Note that other acids may be used for pH control other than the
This is a common colorimetric test for phosphate ions in solution, important for environmental analyses. http://slb.faculty.arizona.edu/sites/slb.faculty.arizona.edu/files/152/Phosphate.pdf
Phosphate + ammonium molybdate + ammonium metavanadate ----> colored complex.
Phosphate(V) ions react with ammonium molybdate to produce a coloured complex.
http://www.rsc.org/learn-chemistry/content/filerepository/CMP/00/001/176/RSC RSC Phosphate by molybdate assay student.pdf

