How do I calculate the bond order for H2- and H2+?
1 Answer
Apr 1, 2017
I'm assuming you mean
Each hydrogen atom contributes one
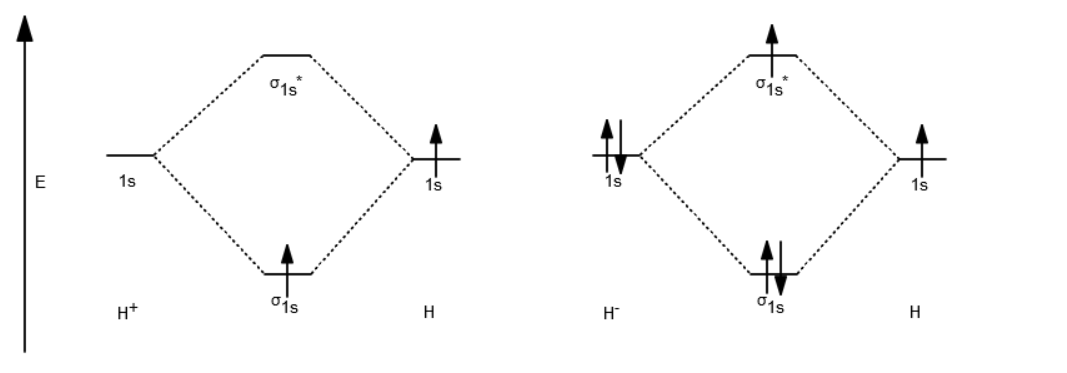
If you calculate their bond order, you get:
#"BO"_(H_2^(+)) = 1/2("Bonding" - "Antibonding")#
#= 1/2(1-0) = 1/2#
#"BO"_(H_2^(-)) = 1/2("Bonding" - "Antibonding")#
#= 1/2(2-1) = 1/2#
So, neither is more stable than the other. But of course, they are less stable than

