Question #f7751
1 Answer
Here's my explanation.
Explanation:
The formula for potassium aluminium sulfate is
It dissociates completely in water according to the equation
We know that
Like many metal cations,
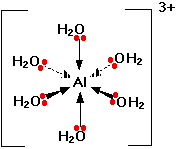
Six water molecules are strongly attached to the aluminium ion by coordinate covalent bonds.
The high positive charge on the aluminium cation pulls electron density toward itself and weakens the adjacent bonds.
Thus, it is easy for a water molecule to remove an
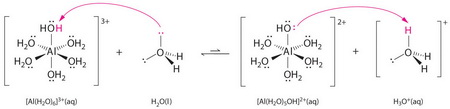
(From saylordotorg.github.io)
The solution becomes acidic.
The
The

