Why are tetrahedral complexes generally high spin?
1 Answer
Sep 14, 2017
Tetrahedral complexes, with
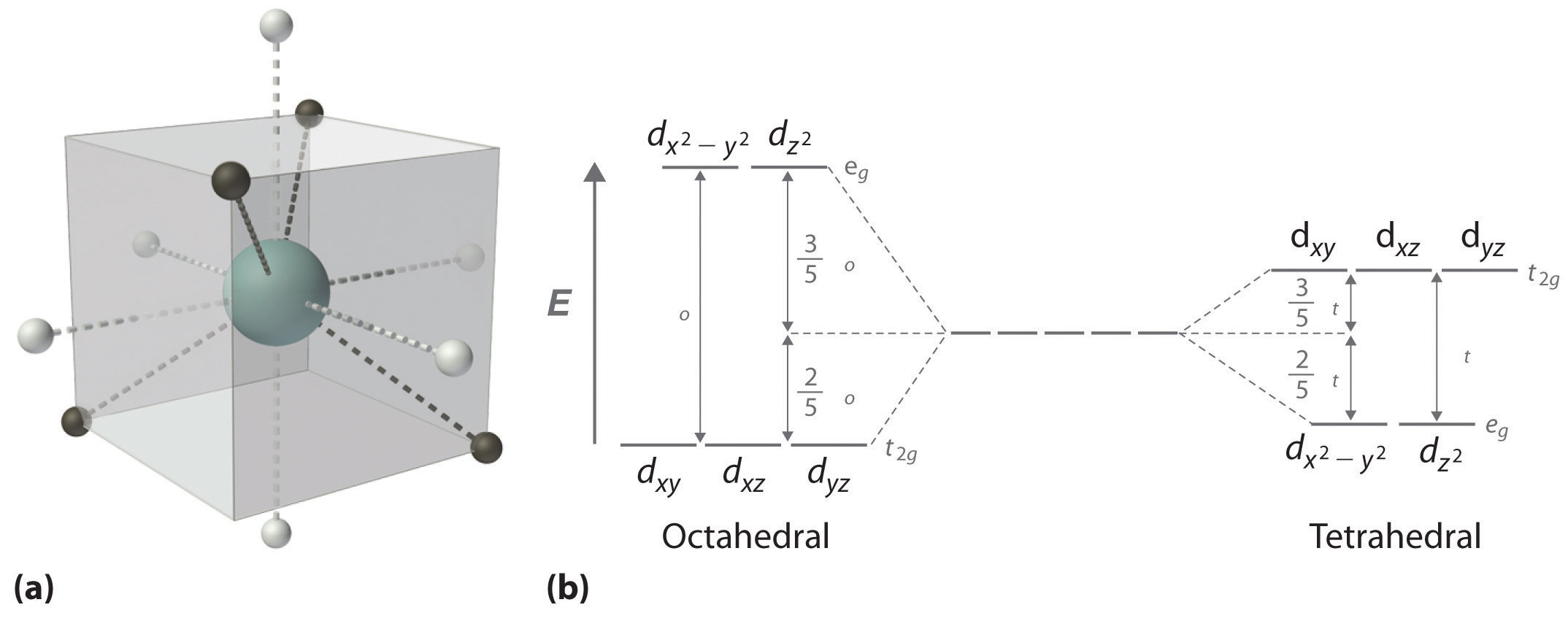
As a result, even with strong-field ligands, the splitting energy is generally smaller than the electron pairing energy.
When electron pairing energy is large, electron pairing is unfavorable. What that implies is that generally, high spin is favored.

