A piece of metal weighs #3.94# #g# and has a density of #5.674# #g##/##cm^3#. What's the volume?
1 Answer
The volume is
Explanation:
The formula for density is
Given
Unknown
Solution
Rearrange the equation to isolate
Remember when you divide by a fraction like
The Density Pyramid below can help you remember how to solve density problems. Cover the variable for which you need to solve. The placement of the known variables on the pyramid will indicate how to solve.
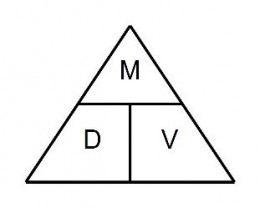
For example, if you need density, cover D. M is over V, so you divide Mass by Volume.
If you need volume, cover V. M is over D, so you divide Mass by Density.
If you need mass, cover M. D is next to V, so multiply Density times Volume.

