Do epinephrine and norepinephrine exist in the body as a racemic mixture? How does the chirality of the compound affect the receptors? Do s-norepinephrine and r-norepinephrine function the same in the human body?
1 Answer
Feb 9, 2016
Natural epinephrine is (
Explanation:
The drugs, however, are often supplied as racemic mixtures.
According to theory, the chiral molecule interacts with three sites on the receptor.
- an anionic site for the ammonium ion
- a hydrogen-bonding site for the β-hydroxyl group
- a flat site for π-π interaction with the aromatic ring
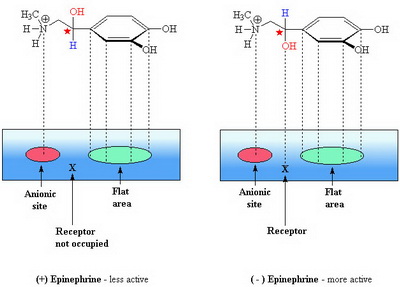
(from tigger.uic.edu)
The less active isomer can establish only a two-point contact.
This loss of the H-bonding interaction equals about 12 kJ/mol, and this corresponds to a 100-fold decrease in activity.
The only difference between epinephrine and norepinephrine is that the latter lacks an N-methyl group.
This does not affect the three-point connection to the receptor.
(

