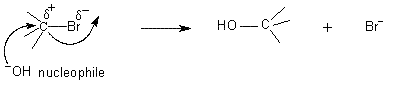Explain nucleophilic substitution of alkyl halides?
1 Answer
Jan 10, 2015
Nucleophilic substitution of alkyl halides is the substitution of the halogen by another group called a nucleophile.

In most of the alkyl halides, the C-X bond is polar.
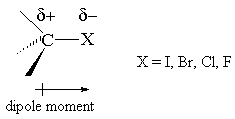
That gives the α carbon a partial positive charge
The positive charge makes that carbon susceptible to attack by a nucleophile.
In one scenario, called the
In a different scenario, the

The nucleophile attacks the carbocation in a second step, forming the product.
Here's a video on the nucleophilic substitution of alkyl halides.