What are common mistakes students make with nucleophilic substitutions?
1 Answer
- Drawing arrows in the wrong order.
- Picking substrates that are too bulky for
#"S"_N2# . - Picking solvents that deactivate the nucleophile.
- Forgetting any possible stereochemical inversions, if applicable.
1) This is more of a pre-emptive concern, but drawing mechanism arrows has a certain flow to it.
It doesn't start at the end, jump to the beginning, then the middle, and back towards the end again. It starts from the beginning and goes all the way to the end.
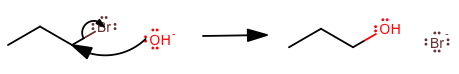
If you drew the upper arrow first, you would be saying that the
2) Many students don't realize that many reactions give a mixture of products. It's not always
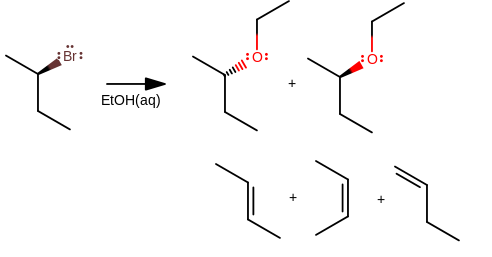
Depending on the conditions, due to the bulkiness of the secondary alkyl halide, you can get any of those products.
- The two stereoisomers are the
#"S"_N1# products. - You might get some
#"S"_N2# if some of the water in solution equilibrates with ethanol to give the ethanolate anion. - Under high heat, you might even get the elimination products shown below the substitution products (likely
#E1# , due to the weak basicity of ethanol).
As a result, we should realize that picking a secondary alkyl halide for
3) Solvents are also quite important. If you want to do an
You should notice that the right mechanism is what you expect to see, generating your product, pentane, and retaining your solvent, ethanol.
The left mechanism, however, is what you don't want to see: the deactivation of your expensive propyllithium nucleophile. Actually, the deprotonated solvent (ethanolate) then acts as the nucleophile, and you don't get your pentane product; instead, you get diethyl ether and a propane side product. Ugh.
In the end, both mechanisms can occur at the same time, giving you that ugly mixture of products, when really, the goal was to make pentane...
4)
As a result, the nucleophile that attacks from above generates a different stereochemistry than the one that attacks from below.

