How are Van der Waals forces and ionic bonds similar?
1 Answer
Aug 1, 2017
They're not that similar...
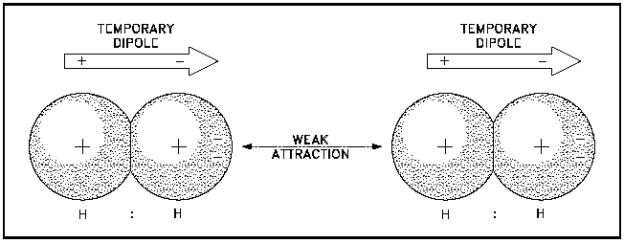
van der Waals forces are short-range intermolecular forces, specifically the interaction of loose, fluctuating PARTIAL charges, i.e. temporary dipoles... dynamic in time, influenced by instantaneously-changing charge density distributions.
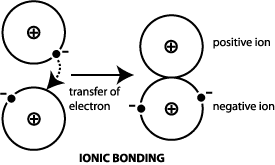
Ionic bonds are interactions of FULL charges, i.e. due to complete transfer of valence electrons. They are permanent charges, and mostly static in time.
NOTE: Ionic bonding should not be confused with ion-pairing interactions:
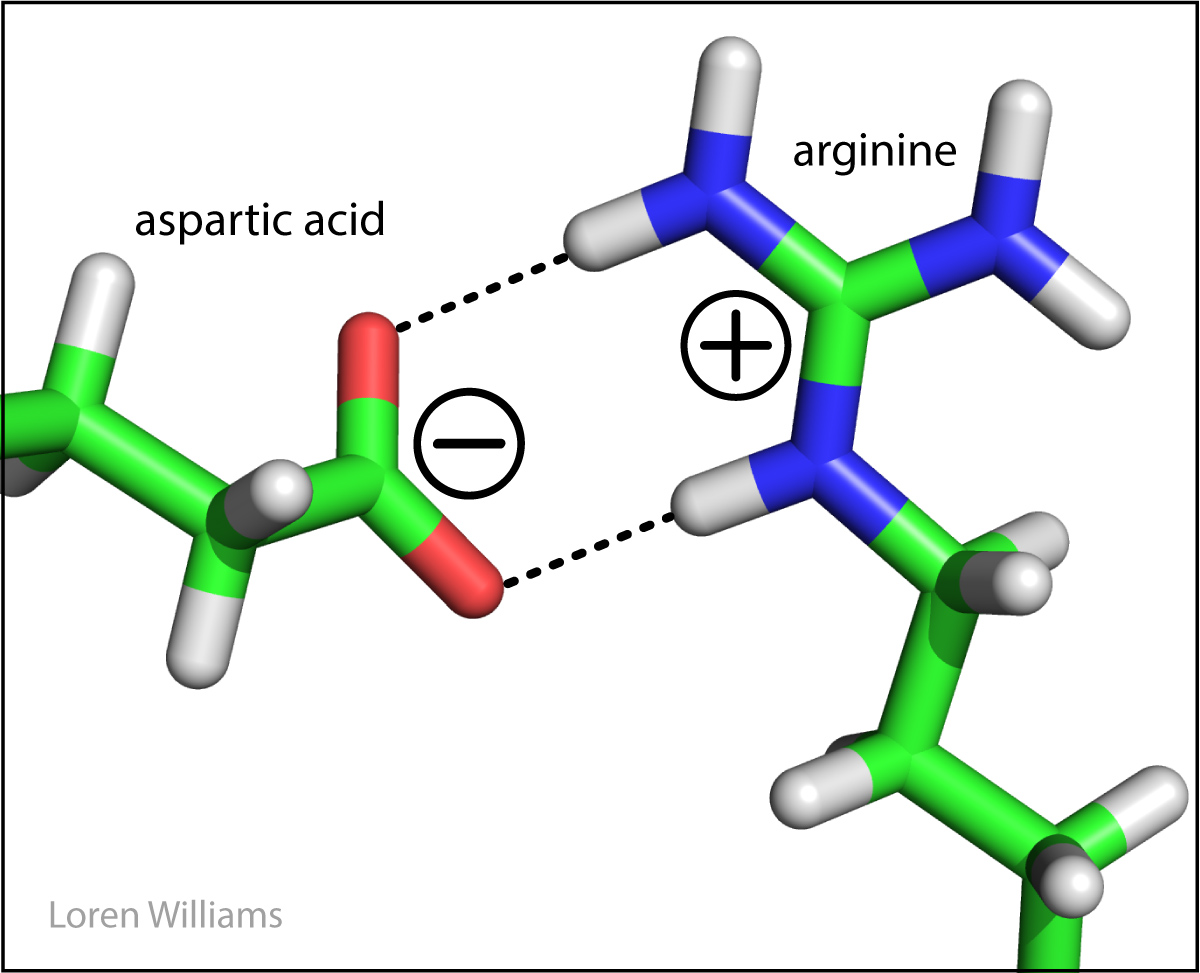
Aspartate and arginine are clearly not bonding to each other, but their full charges are still interacting.

