How can I predict the bond angles for IF_4""^(-)?
1 Answer
You examine the molecular and electron-domain geometries of the ion.
Explanation:
First, start by drawing the Lewis structure for the
The
The iodine atom will be the central atom. It will form four single bonds with the fluorine atoms, for a total of 8 out of the 36 valence electrons available.
Each of the four fluorine atoms will have 3 lone pairs of electrons attached, which brings thte total number of valence electrons used to 32.
The remaining 4 valence electrons will be placed on the the iodine atom as lone pairs.
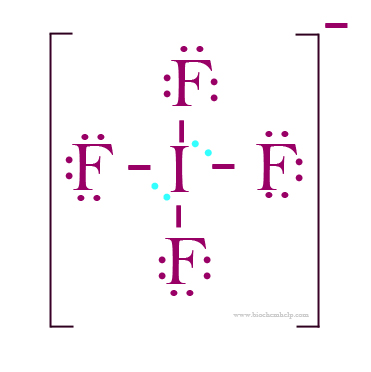 http://biochemhelp.com/IF4-lewis-structure.html
http://biochemhelp.com/IF4-lewis-structure.html
The central atom is surrounded by 6 regions of electron densiy - 4 single bonds and 2 lone pairs. This means that its steric number is equal to 6.
Its coordination number, which is given by the number of atoms that are bonded to iodine, is equal to 4.
The molecular geometry of the ion, which does not take into account the lone pairs present on the central atom, will be square planar,
The electron-domain arrangement, which does include any lone pairs attached to the central atom, will be octahedral.
http://pt.slideshare.net/marsjomm/unidade-4-geometria-molecular
As you can see in the above image, the two lone pairs present on the central atom occupy possitions opposite to each other.
This configuration actually provides the minimum repulsion between the lone pairs and the bonding electrons. More specifically, the repulsion coming from the lone pairs cancels out, since one is pressing down and the other one is pressing up on the bonding electrons.
This means that you can expect the bond angles of the molecule to be very close to ideal.
For a square planar molecule, ideal bond angles measure

