How can #"E/Z"# isomers arise?
1 Answer
Feb 2, 2017
Consider the simple molecule
Explanation:
Now for
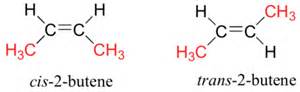
For both isomers, the connectivity is MANIFESTLY the same:
For example,
Consider the simple molecule
Now for

For both isomers, the connectivity is MANIFESTLY the same:
For example,