How do you compare ionic radii?
1 Answer
Jun 9, 2018
Well, you have to have some data first....
Explanation:
And I have used this illustration before....
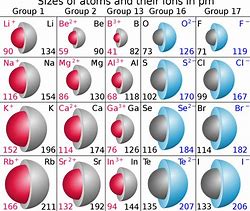
The units are in
Well, you have to have some data first....
And I have used this illustration before....

The units are in