How do you draw an atomic level diagram for 2S+3O2=2SO3 ?
1 Answer
I think you are asking how to show the way the atoms combine in the reaction.
Explanation:
The easiest way is first to break up the
#"O"_2 → 2"O"#
Then we can re-write the equation for the reaction as
#"2S + 6O → 2SO"_3#
or
#"1S + 3O → 1SO"_3#
The
If we want to show how the atoms combine, the diagram might look like this:
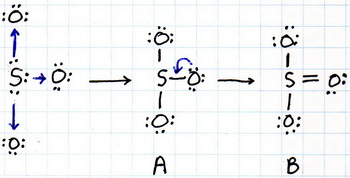
Structure A isn't a good structure, because it doesn't give the
So we use a pair of electrons from an
Structure B still isn't the best structure for
If this isn't what you wanted, please let me know.

