How many electrons are in an atom that has an electron configuration of 2,8,8?
1 Answer
Explanation:
All you really have to do here is add up the number of electrons given to you for each shell of the atom.
As you know, electrons that surround the nucleus are located in energy shells, or energy levels.
Each energy level can hold a maximum number of electrons that depends on how many subshells, and subsequently orbitals, that energy level can hold.
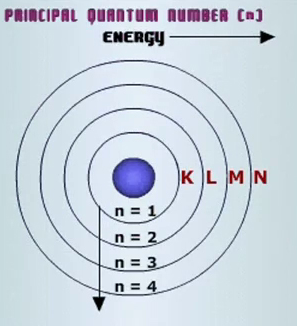
In your case, only the first three energy shells are of interest. You have
- the first energy level, or
#"K"#
This energy level has one subshell, the s-subshell, and one orbital, the s-orbital.
- the second energy level, or
#"L"#
This energy level has two subshells, the s-subshell and the p-subshell, and a total of four orbitals, one s-orbital and three p-orbitals.
- the third energy level, or
#"M"#
This energy level has three subshells, the s-subshell, the p-subshell, and the d-subshell, and a total of nine orbitals, one s-orbital, three p-orbitals, and five d-orbitals.
Now, each orbital can hold a maximum of two electrons. This means that each energy shell can hold a maximum of
#"K: " 2 xx "1 orbital" = "2 electrons"#
#"L: " 2 xx "4 orbitals" = "8 electrons"#
#"M: " 2 xx "9 orbitals" = "18 electrons"#
And so on. In your case, the element is said to contain
The total number of electrons present in the atom will thus be equal to
#"no. of e"^(-) = 2 + 8 + 8 = color(green)(|bar(ul(color(white)(a/a)"18 e"^(-)color(white)(a/a)|)))#