How many unpaired electrons in a ground state fluorine atom?
1 Answer
Jul 21, 2016
Fluorine has one unpaired electron in the ground state.

Explanation:
Fluorine is in the
The outermost shell of the atom would have

Image from @smarterteacher
The Bohr Model would have

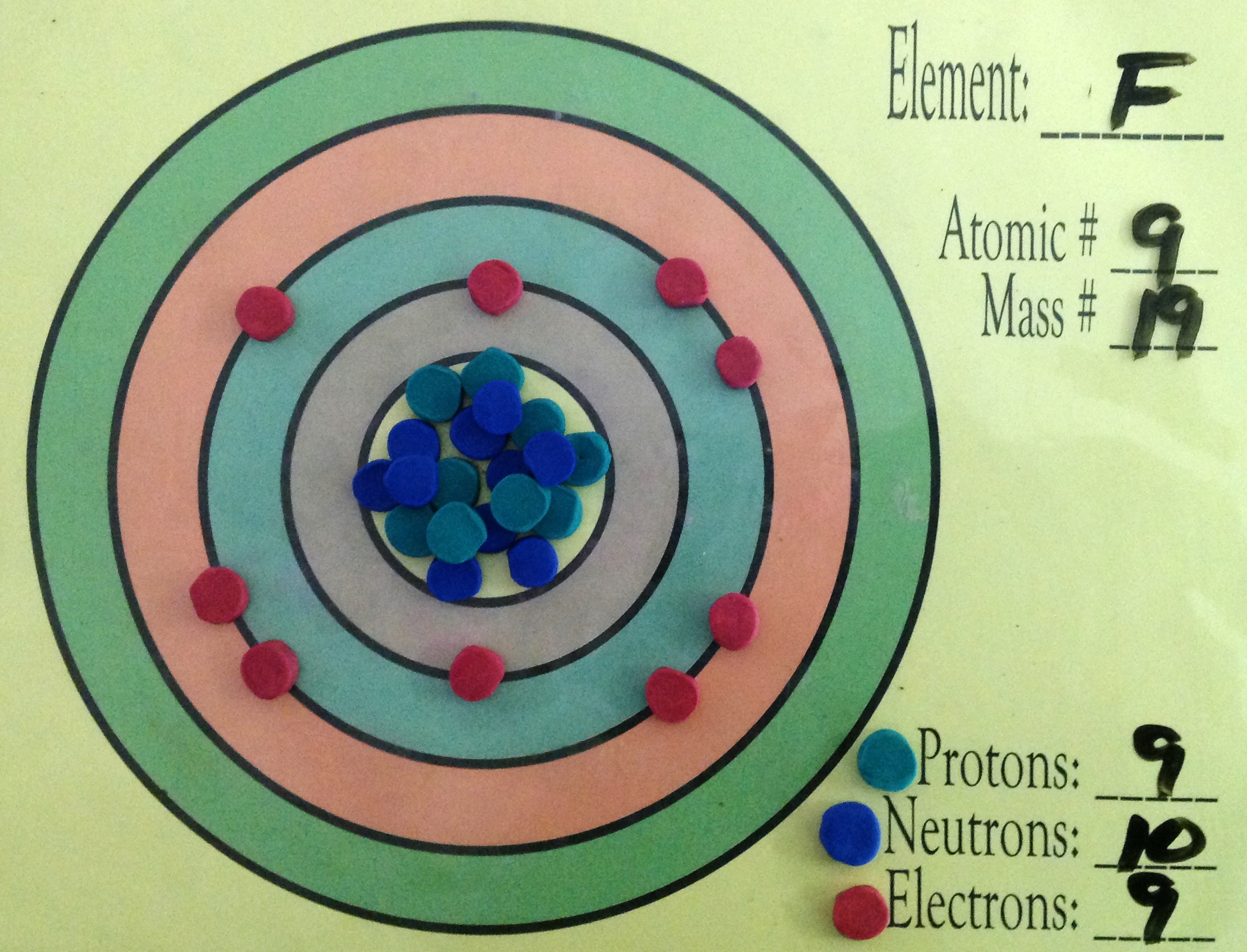
Images from @smarterteacher

