How many valence electrons does cadmium have?
1 Answer
May 11, 2018
Explanation:
Here's cadmium on the periodic table:
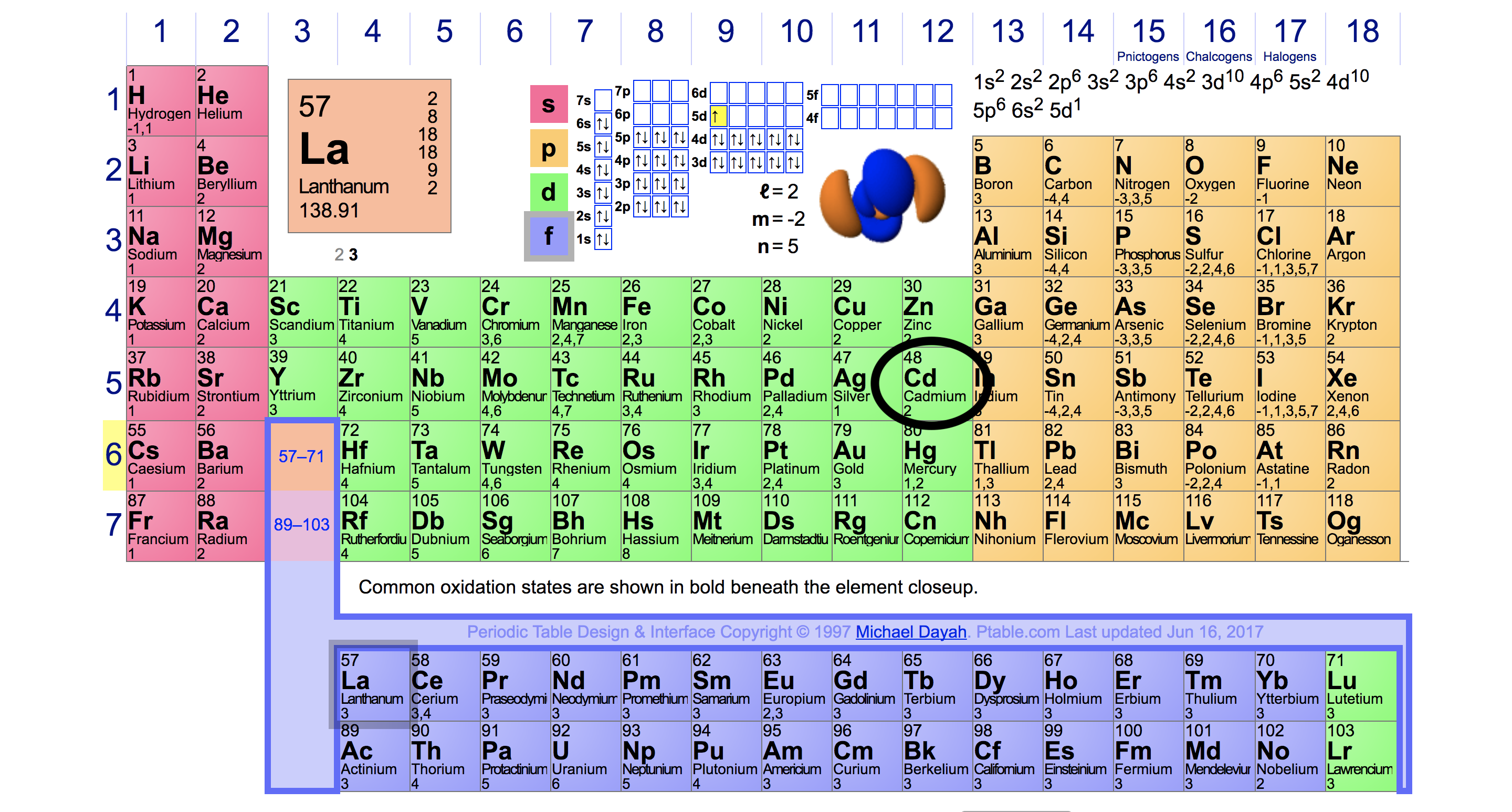
Its electron configuration would be
So, as with most transition metals, it will have

