How would you write the equation for this reaction: excess potassium hydroxide solution is added to a solution of potassium dihydrogen phosphate?
1 Answer
Dec 3, 2016
Explanation:
This is simply an acid base reaction. It is tempting to speculate that the addition of excess hydroxide would drive the equilibrium to the right, and result in the formation of
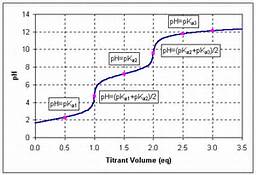
How does the graph illustrate what I have said?

