If the pressure on a gas is decreased by one-half, how large will the volume change be?
1 Answer
Jun 13, 2017
The volume will increase by a factor of
Explanation:
Boyle's Law states that the pressure of a gas is inversely proportional to its volume, assuming that all other variables are constant.
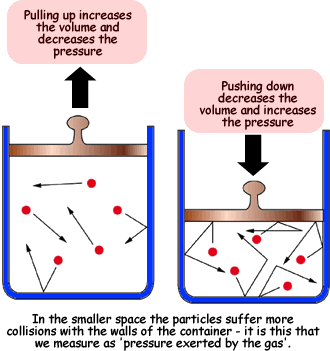
Modeled by the equation:
where:
#P# is the pressure in whatever units you desire. The units must be used consistently. I prefer atmospheric pressure .
#V# is the volume of the gas in whatever units you desire. The units must be used consistently. I prefer litres.- the subscripts
#1# and#2# are before and after the change, respectively.
Due to the inverse proportion of the pressure of the gas and its volume, if the pressure was decreased to
Hope this helps :)


