How do I find the eigenvalues for the finite potential well of width 2L?
Derive a transcendental equation for the allowed energies and solve it graphically in the finite well of the height #V_0#
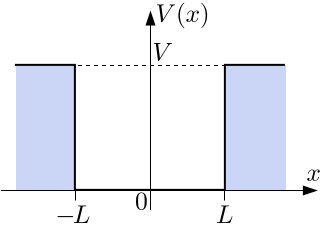
#V(x) = {(-V_0 ,|x|>=L),(0, "elsewhere"):}#
#alphatanalpha = k# (even parity)
#alphacotalpha = -k# (odd parity)
i hope that u understand what #alpha and k# are.
but to mark the energy eigenvalues
i had to make circle of radius #alpha + k#
what is the physical reason behind this?
Derive a transcendental equation for the allowed energies and solve it graphically in the finite well of the height
i hope that u understand what
but to mark the energy eigenvalues
i had to make circle of radius
what is the physical reason behind this?
1 Answer
Here is the Excel sheet I made while doing this.
The radius of the circle just tells you what you set the height of your potential well to be. However, its radius is given by
#lim_(V_0 -> oo) "Finite Well" = "Infinite Potential Well"#
If we choose
#ul(E" "" "" "" "" "" "E//V_0" "" "color(white)(.)"Quantum Number"" "" ")#
#(1.63948ℏ^2)/(2mL^2)" "" "0.081974" "" "1.28042#
#(6.44190ℏ^2)/(2mL^2)" "" "0.322095" "" "2.53809#
#(13.89150ℏ^2)/(2mL^2)" "color(white)(.)0.694575" "" "3.72713#
The graph below utilizes a well width of
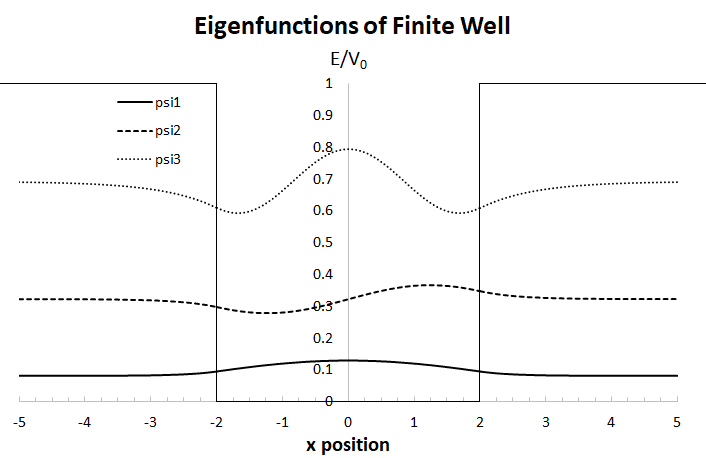
Well, let's start by properly defining the problem... instead of defining a tunnelling problem with a barrier of width
#V(x) = {(V_0, |x| >= L), (0, -L < x < L):}#
That way, we get:
Then, let's define the eigenfunctions for each region
#psi_(I) = Ae^(-alphax) + Be^(alphax), " "" "" "" "-oo < x < -L#
#psi_(II) = Csin(kx) + Dcos(kx), " "-L < x < L#
#psi_(III) = Fe^(-alphax) + Ge^(alphax), " "" "" "" "L < x < oo# where:
#alpha = sqrt((2m(V_0 - E))/ℏ^2)# ,#" "E < V_0# for bound states
#k = sqrt((2mE)/ℏ^2)#
Since the wave function must vanish at
EVEN SOLUTIONS
For the even solutions, the
#psi_(I) = Be^(alphax), " "" "" "-oo < x < -L#
#psi_(II) = Dcos(kx), " "-L < x < L#
#psi_(III) = Be^(-alphax), " "" "" "L < x < oo#
Now we apply the continuity conditions.
#ul(psi_(I)(-L) = psi_(II)(-L))# :
#Be^(-alphaL) = Dcos(-kL)# #" "" "" "bb((1))#
#ul((dpsi_(I)(-L))/(dx) = (dpsi_(II)(-L))/(dx))# :
#alphaBe^(-alphaL) = -kDsin(-kL)# #" "bb((2))#
#ul(psi_(II)(L) = psi_(III)(L))# :
#Dcos(kL) = Be^(-alphaL)# #" "" "" "" "bb((3))#
#ul((dpsi_(II)(L))/(dx) = (dpsi_(III)(L))/(dx))#
#kDsin(kL) = alphaBe^(-alphaL)# #" "" "" "bb((4))#
Taking
#color(green)(alpha = ktan(kL))#
Now, let
From the definitions of
#u_0^2 -= u^2 + v^2 = alpha^2L^2 + k^2L^2#
#= (2mL^2(V_0 - E))/ℏ^2 + (2mL^2E)/ℏ^2 = (2mL^2V_0)/ℏ^2#
So now for the even solution, we can graph
#alphaL = kLtan(kL)#
or
#u = sqrt(u_0^2 - v^2) = vtanv#
Now we can simply use Excel to graph
The intersections give the quantum numbers for each eigenvalue.
Suppose
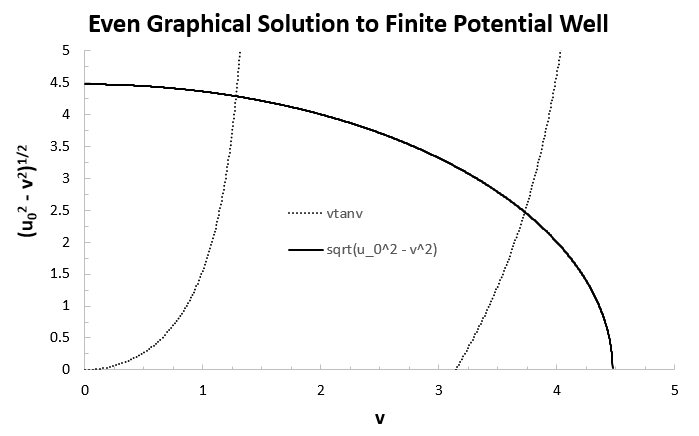
For the even solutions we got
For now, each eigenvalue is given in terms of the quantum number
#v_n^2 = k_n^2L^2 = (2mL^2E_n)/ℏ^2#
#=> color(green)(E_n = (ℏ^2v_n^2)/(2mL^2))#
Now let's move on to the odd solutions.
ODD SOLUTIONS
Now the origin is a rotation axis, so that
#psi_(I) = Be^(alphax), " "" "" "-oo < x < -L#
#psi_(II) = Csin(kx), " "-L < x < L#
#psi_(III) = -Be^(-alphax), " "" "L < x < oo#
Now we apply the continuity conditions as before.
#ul(psi_(I)(-L) = psi_(II)(-L))# :
#Be^(-alphaL) = Csin(-kL)# #" "" "" "bb((1))#
#ul((dpsi_(I)(-L))/(dx) = (dpsi_(II)(-L))/(dx))# :
#alphaBe^(-alphaL) = kCcos(-kL)# #" "" "bb((2))#
#ul(psi_(II)(L) = psi_(III)(L))# :
#Csin(kL) = -Be^(-alphaL)# #" "" "" "bb((3))#
#ul((dpsi_(II)(L))/(dx) = (dpsi_(III)(L))/(dx))#
#kCcos(kL) = alphaBe^(-alphaL)# #" "" "" "bb((4))#
Taking
#color(green)(alpha = -kcot(kL))#
Using the same substitutions as before, we again graph
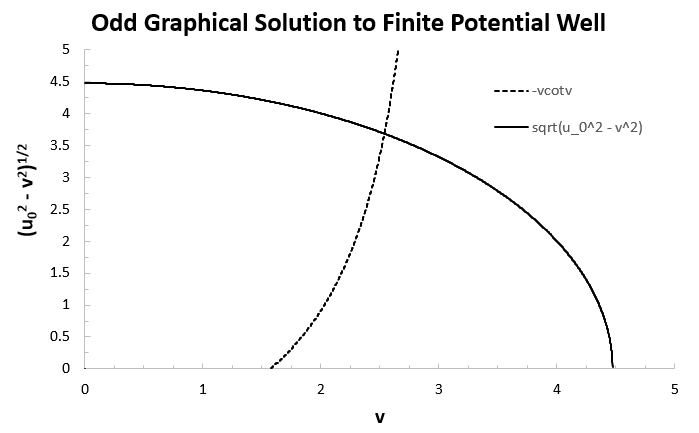
In this case our well only had three energy levels. Here we again had
COMBINED SOLUTIONS
And combining them onto one graph, we get the three energy levels in this finite well with
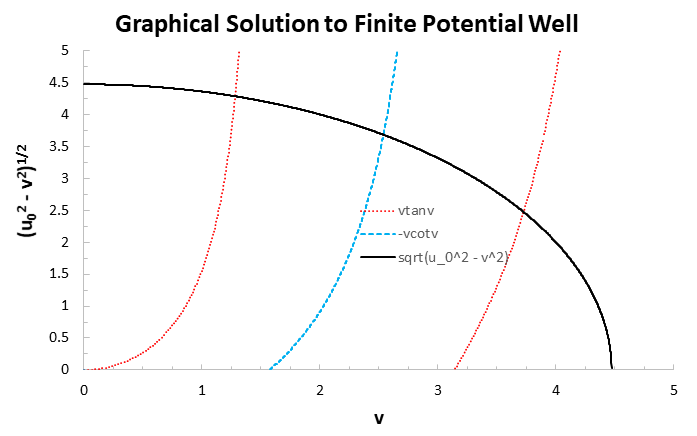
#color(blue)(E_1 = (1.63948ℏ^2)/(2mL^2))#
#color(blue)(E_2 = (6.44190ℏ^2)/(2mL^2))#
#color(blue)(E_3 = (13.89150ℏ^2)/(2mL^2))#
And of course, if we let
The choice of

