The surface tension of benzene at 20°C is 28.85 dyne/cm. In a capillary apparatus the liquid rose to a height of 1.832 cm. The density of benzene is 0.8765 g/mL. How would you calculate the diameter of the capillary tube in mm?
1 Answer
The diameter of the capillary tube is 0.366 mm.
Explanation:
One method for measuring the surface tension of a liquid is to measure the height the liquid rises in a capillary tube.
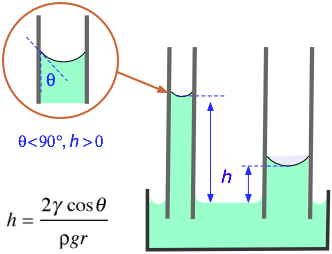
The formula is
#color(blue)(|bar(ul(color(white)(a/a) h = "2γcosθ"/"ρgr" color(white)(a/a)|)))" "#
where
We can rearrange the equation to get
#color(blue)(|bar(ul(color(white)(a/a) r = "2γcosθ"/(hgρ)color(white)(a/a)|)))" "#
For most liquids and clean glass, the contact angle
If
#color(blue)(|bar(ul(color(white)(a/a) r = "2γ"/(hgρ)color(white)(a/a)|)))" "#
In your problem,

